Trending Stories
“Decoding Vaccine Adverse Event Reporting in Canada: Analyzing Health Canada’s Delayed Two-Year Response”

Decoding Vaccine Adverse Event Reporting in Canada: Analyzing Health Canada’s Delayed Two-Year Response”
Spring of 2021: Confusion Brews
In 2021, health professionals and consumers voiced their confusion about how to effectively report AEFIs. It took Health Canada almost two years to address these concerns, highlighting potential bottlenecks in the regulatory structure of public health services.
The issue was further underscored by the significant increase in the reports of serious adverse events related to COVID-19 vaccinations compared to the average AEFI report count.
February 2023: A Reinstated Solution
In a surprising turn of events, Health Canada reintroduced a direct link to its online reporting form in February 2023, two years after removing it in December 2020. This move happened around the same time that the novel COVID-19 injections were authorized under emergency use legislation.
Conservative MP Colin Carrie openly questioned the federal regulatory agency about this change, highlighting potential discrepancies in communication and documentation of adverse events, particularly involving vaccine-related injuries.
Two Systems, One Purpose: The Reporting Complication
Intricacies arise when two separate systems exist for adverse events reports – the Canadian Adverse Events Following Immunization Surveillance System (CAEFISS) managed by the Public Health Agency of Canada (PHAC), and Health Canada‘s own Canada Vigilance Program (CVP).
Both systems work together in tracking and documenting adverse events from all marketed health products, including vaccines.
However, the presence of two systems, coupled with certain exemptions for hospitals reporting AEFIs through provincial/territorial systems, has led to operational confusion. For example, emergency room physicians found it challenging to report COVID-19 vaccine reactions due to bureaucratic red tape.
The Road Ahead: Addressing Under-reporting and Improving Stakeholder Communications
The confusion that Health Canada allowed to persist for two years may have had far-reaching implications. From the potential under-reporting of adverse events to the struggles faced by those suffering from vaccine injuries to get their reactions formally documented for compensation, the challenges are myriad.
As we navigate this unprecedented global health crisis, it is imperative to prioritize transparent communication, efficient reporting systems, and quick responses to stakeholder concerns. Only then can we ensure the credibility and effectiveness of our public health initiatives.
Reference Links:
source Rebel News Article
Trending Stories
Sister Regina Liu: Empowering Health Through Acupuncture

Sister Regina Liu: Empowering Health Through Acupuncture
In the bustling world of healthcare, Sister Regina Liu stands out as a beacon of holistic healing. Her journey into the world of acupuncture is not only inspiring but also transformative for the countless individuals she has treated.
Through her dedication, Sister Regina has brought traditional Chinese medicine to the forefront, offering an alternative and complementary approach to modern medical practices.
The Journey of Sister Regina Liu
Sister Regina Liu’s path to becoming a renowned acupuncturist began with her deep-rooted interest in holistic health. Born into a family that valued traditional Chinese medicine, Sister Regina was exposed to the benefits of acupuncture from a young age. Her early fascination turned into a lifelong passion as she pursued formal education and training in the field.
Acupuncture: Bridging Ancient Wisdom and Modern Health
Acupuncture, a practice with origins in ancient China, involves inserting thin needles into specific points on the body to balance the flow of energy or “qi.” Sister Regina Liu has mastered this ancient art, using it to address a wide range of health issues.
From chronic pain to stress management, her expertise has provided relief to many who had exhausted conventional treatment options.
Impact on Community Health
Sister Regina’s impact extends beyond individual treatments. She has been instrumental in educating the community about the benefits of acupuncture, breaking down misconceptions, and making the practice more accessible.
Her workshops and seminars have enlightened many about the holistic approach to health, emphasizing the interconnectedness of body, mind, and spirit.
Success Stories and Testimonials
The success stories of Sister Regina’s patients are a testament to her skill and dedication. Many individuals who had lost hope found solace in her treatments.
For instance, Maria, a long-time sufferer of migraines, experienced significant relief after just a few sessions with Sister Regina. Her story is just one of many that highlight the transformative power of acupuncture under Sister Regina’s care.
Challenges and Triumphs
Like any journey, Sister Regina’s path was not without challenges. Integrating acupuncture into mainstream healthcare faced resistance initially.
However, her perseverance and the undeniable results of her treatments gradually won over skeptics. Today, Sister Regina is not only respected in the field of acupuncture but also in the broader medical community.
The Science Behind Acupuncture
While acupuncture is rooted in ancient practices, modern science has begun to unravel the mechanisms behind its effectiveness. Studies have shown that acupuncture can stimulate the release of endorphins, the body’s natural painkillers, and improve blood circulation.
These scientific validations have further cemented acupuncture’s place in contemporary healthcare, thanks in part to advocates like Sister Regina Liu.
Acupuncture in Modern Healthcare
Sister Regina’s work exemplifies how traditional practices can complement modern medicine. Hospitals and clinics increasingly incorporate acupuncture into their treatment plans, recognizing its benefits in pain management, mental health, and overall well-being. This integration signifies a broader acceptance and understanding of holistic health practices.
Future Vision
Looking ahead, Sister Regina Liu envisions a future where acupuncture and traditional Chinese medicine are fully integrated into the global healthcare system. She continues to advocate for research, education, and policy changes that support the inclusion of holistic practices in mainstream medicine.
How to Get Started with Acupuncture
For those new to acupuncture, Sister Regina offers practical advice on getting started. She recommends finding a certified acupuncturist, understanding the treatment process, and maintaining an open mind. Her guidance helps demystify acupuncture, making it more approachable for newcomers.
Conclusion
Sister Regina Liu’s journey in empowering health through acupuncture is a remarkable tale of dedication, resilience, and success. Her contributions have not only alleviated individual suffering but also enriched the broader understanding of holistic health. As acupuncture continues to gain recognition, Sister Regina’s legacy will undoubtedly inspire future generations of healers.
FAQs
1. What conditions can acupuncture treat?
Acupuncture can address various conditions, including chronic pain, migraines, stress, anxiety, digestive issues, and more. It is also used to support overall wellness and balance.
2. Is acupuncture safe?
Yes, when performed by a certified and experienced acupuncturist, acupuncture is safe. It involves using sterile, single-use needles and adhering to proper hygiene practices.
3. How many sessions are needed to see results?
The number of sessions varies depending on the condition and individual response. Some may experience relief after one session, while others may need multiple treatments.
4. Does acupuncture hurt?
Acupuncture needles are very thin, and most people feel minimal to no discomfort. Some may feel a slight tingling or warmth at the needle site.
5. How do I find a qualified acupuncturist?
Look for acupuncturists who are certified by recognized professional organizations and have positive patient reviews. Personal recommendations and consultations can also help in making an informed choice.
References
Trending Stories
In 2 Shape Gym Unveils Major Expansion in Stourport
Trending Stories
9 Reasons Why In-Person Friendships Are Irreplaceable
-

 Trending Stories1 year ago
Trending Stories1 year agoCDC: 1 in 4 Americans Still COVID-Free by End of 2022
-

 Health5 years ago
Health5 years agoMeghan Trainor Shares Motivational New Song ‘Blink’
-
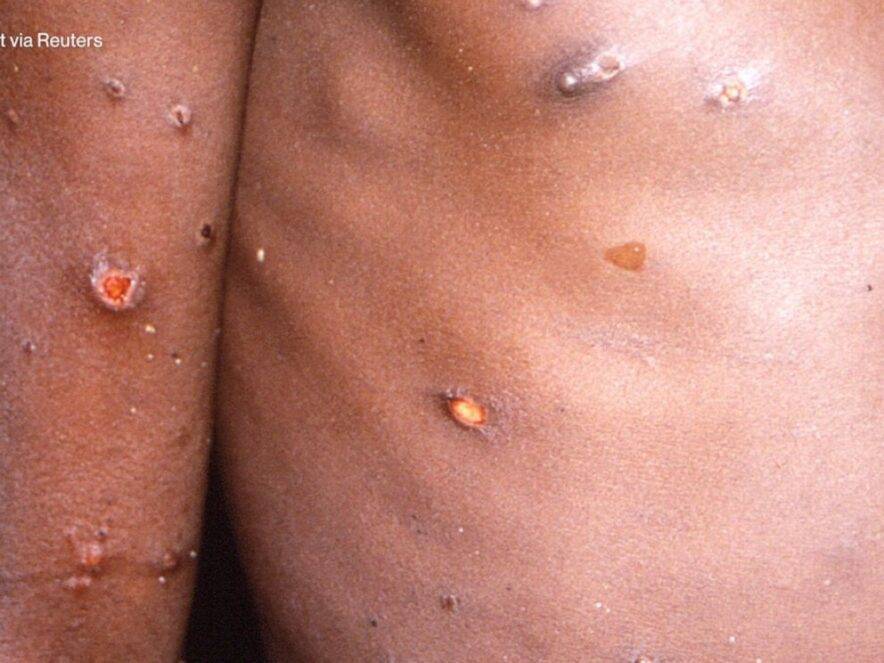
 Health2 years ago
Health2 years agoHow Long Does Monkey Pox Last Before It Surfaces in the Body?
-

 Health2 years ago
Health2 years agoWhat Causes Swollen Body? Understanding Edema and its Triggers
-

 Health3 years ago
Health3 years agoNutrition and the Importance of a Fitness Program – 3 Things to Know
-

 Health3 years ago
Health3 years ago5 Weird Reasons Why Pimples Disappear After Marriage
-

 Health3 months ago
Health3 months agoHow Do Pawpaw Seeds Support Cardiovascular Health?
-

 Health2 years ago
Health2 years agoHealth Benefits Of Pawpaw Seed? 7 Things To Know